NIH-funded consortium focuses on advancing genome editing research
The Somatic Cell Genome Editing Consortium (SCGE), a massive research effort funded by the National Institutes of Health, aims to accelerate genome editing research and the development of new gene-editing related technologies and therapeutic approaches. The collaborative initiative includes 45 projects from 38 institutions and 72 principal investigators, including researchers from Baylor College of Medicine and Rice University. A collaborative manuscript published in Nature describes the overall organization, goals and planned activities.
SCGE-funded groups are engaged in the development of new delivery systems for genome editing machinery, identification of new editing enzymes, assessing the risks of genome editing and generating reporter models for detecting editing events, as well as testing in small- and large-animal models to support these efforts.
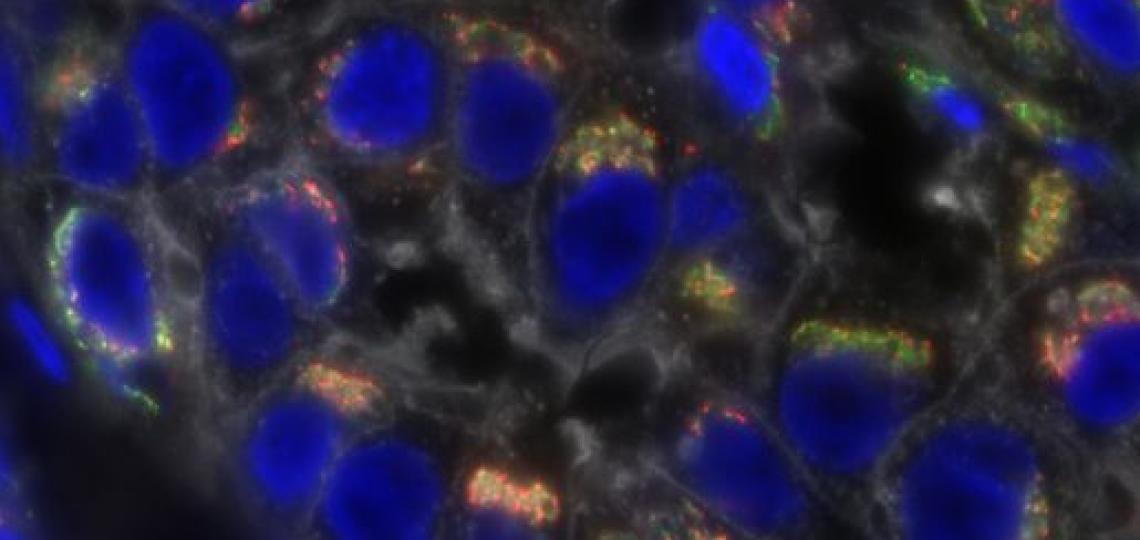
A Baylor team led by Dr. Jason Heaney, Dr. William Lagor and Dr. Mary Dickinson is one of two groups that serve as a small-animal testing center. They are responsible for developing new reporter mice to detect somatic genome editing events. The mouse models that are being developed by Heaney’s lab allow for detection of genome editing events through activation of fluorescence and luminescent reporter genes. The new models can detect different types of genome editing events, including small insertion and deletion mutations, homology directed repair and off-target editing. Dr. Denise Lanza and Dr. John Seavitt are leading the mouse production efforts.
“Measuring these editing events in live animals has been a major challenge until this point,” said Heaney, associate professor of molecular and human genetics and director of the Center for Precision Medicine Models at Baylor. “The new animal models allow for identification of rare individual cells that have undergone genome editing. For example, the models now make it possible to test whether new delivery systems could enter unintended tissues, including germ cells.”
Dickinson’s lab will coordinate research activities for the grant, including developing and applying new imaging modalities to quantify genome editing with the assistance of Dr. Logan Hsu, assistant professor of molecular physiology and biophysics. Sophisticated imaging modalities such as light sheet microscopy and tissue clearing are being used to identify editing events with single-cell resolution within the reporter mice.
Lagor’s lab serves as the genome editing testing core, which is tasked with validation of the newly generated reporter mice. This includes delivery of genome editing enzymes such as the CRISPR/Cas9 system. As part of the SCGE, the small-animal testing centers also provide independent validation for teams developing new delivery systems. This effort to ensure rigor and reproducibility involves independent validation of new delivery technologies in the reporter mice at Baylor. Dr. Christopher Walkey in the Lagor group coordinates with 10 different teams from across the U.S. to test their novel delivery systems, which include everything from engineered viruses, to peptides, to lipid nanoparticles. Ayrea Hurley and Alexa Martinez are testing these delivery systems in the new reporter mice.
Dr. Gang Bao, Foyt Family Professor and chair of the Department of Bioengineering at Rice University, is an expert in CRISPR/Cas9 based genome editing, including the prediction and quantification of genome editing outcomes. In addition to supporting the efforts of the small-animal testing center at Baylor, Bao leads a delivery group in the SCGE. This is a collaborative project with Lagor, associate professor of molecular physiology and biophysics at Baylor, Dr. Joshua Wythe, associate professor of molecular physiology and biophysics at Baylor, Dr. Douglas Burrin, research physiologist and professor of pediatrics at Baylor and the USDA-ARS Children’s Nutrition Research Center, and Dr. Xue Sherry Gao, assistant professor in chemical and biomolecular engineering at Rice. Bao and his collaborators have been working on engineering adeno-associated viruses (AAV) to deliver genome editing enzymes to the endothelium.
“The goal of this SCGE delivery project is to engineer a modular AAV vector platform to enhance tissue-specific delivery and attach Cas9 proteins to AAV to achieve transient gene editing activity,” Bao said.
Dr. Buhle Moyo in the Bao lab has been engineering the AAV capsid to attach the Cas9 protein. Dr. Michele Alves-Bezerra in the Lagor lab is developing new approaches to improve AAV delivery to endothelial cells.
“Delivery of CRISPR/Cas9 to the vasculature remains a major challenge and could have important applications to treat a wide range of diseases including atherosclerotic cardiovascular disease and macular degeneration,” Lagor said.
Results from experiments in the SCGE will become part of a publicly available toolkit that will allow researchers from around the world to benefit from these cutting-edge technologies. Learn more here.