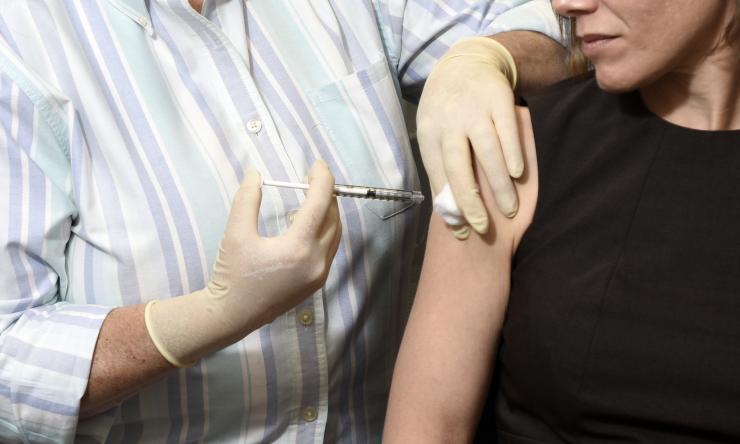
New partnership to accelerate research of COVID-19 vaccine
The National School of Tropical Medicine at Baylor College of Medicine and Texas Children’s Hospital Center for Vaccine Development have formed a new partnership with PATH, a global nonprofit organization dedicated to improving public health. Together, these institutions will advance the development of a low-cost, safe and effective vaccine to prevent COVID-19.
Through this alliance, researchers with Baylor, Texas Children’s and PATH hope to accelerate the vaccine candidate into Phase 1 clinical evaluation later this year. There is an urgent need to advance a COVID-19 vaccine, especially for low- and middle-income nations where diseases often take their greatest toll.
“It is becoming increasingly apparent that this virus poses great risk to low- and middle-income countries of South and Central America, Africa and Asia,” said Dr. Maria Elena Bottazzi, associate dean of the National School of Tropical Medicine at Baylor and co-director of Texas Children’s Hospital Center for Vaccine Development. “Our goal is to ensure that our development efforts lead to COVID-19 vaccines with global access, so populations can benefit in the many low-resource countries where it is so greatly needed.”
“Throughout my 40-year career in science and global health, I’ve long admired the important role of PATH in accelerating vaccines and other global health technologies,” said Dr. Peter Hotez, also co-director of Texas Children’s Hospital Center for Vaccine Development and dean of the National School of Tropical Medicine at Baylor. “It’s exciting to now join in this endeavor with PATH to address this important global health threat.”
PATH has expertise in advancing the development and licensure of affordable and effective vaccines and ensuring their availability and accessibility for low- and middle-income countries, including new vaccines for meningococcal A meningitis, malaria, pneumococcal disease and rotavirus.
Between 2011 to 2016, Baylor and Texas Children’s led a consortium with the New York Blood Center, UTMB Galveston and Walter Reed Army Institute of Research to advance recombinant protein vaccines for severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS) – and now COVID-19. Their platform is a recombinant protein similar to the technology used to produce and distribute recombinant protein hepatitis B vaccine worldwide.
The new alliance will advance the COVID-19 project to generate proof-of-concept safety and immunological data in humans for a vaccine candidate originally developed by this consortium against SARS, but that also may have the ability to prevent COVID-19 because of genetic similarities, cross-protection and neutralization between the SARS and COVID-19 coronaviruses. Material produced according to Good Manufacturing Practice standards already is available for study use, enabling an accelerated pathway to the clinic.
“A vaccine is needed as soon as possible to provide protection against COVID-19, interrupt transmission and prevent future outbreaks globally, but it is especially critical in parts of the world where health systems are weaker and mitigation efforts like hand washing or social distancing are not feasible,” says Deborah Higgins, senior director for PATH’s Center for Vaccine Innovation and Access disease area focused on respiratory infections. “As such, this collaboration is an important step toward understanding this vaccine candidate’s potential as a COVID-19 prevention tool and ensuring that it can be within reach for everyone if successful, no matter where one lives.”
PATH will apply an integrated portfolio and financial management process, providing support to Baylor in project and quality management, the execution of the regulated preclinical studies, engagement with U.S. regulatory agencies, and clinical study design and sponsorship. The clinical study is anticipated to be conducted by and at Baylor. Initial funding for this new alliance, provided by Texas Children’s and Baylor, will take the project through Investigational New Drug submission and Phase 1 planning. Additional funding is being sought for the conduct of the Phase 1 study.