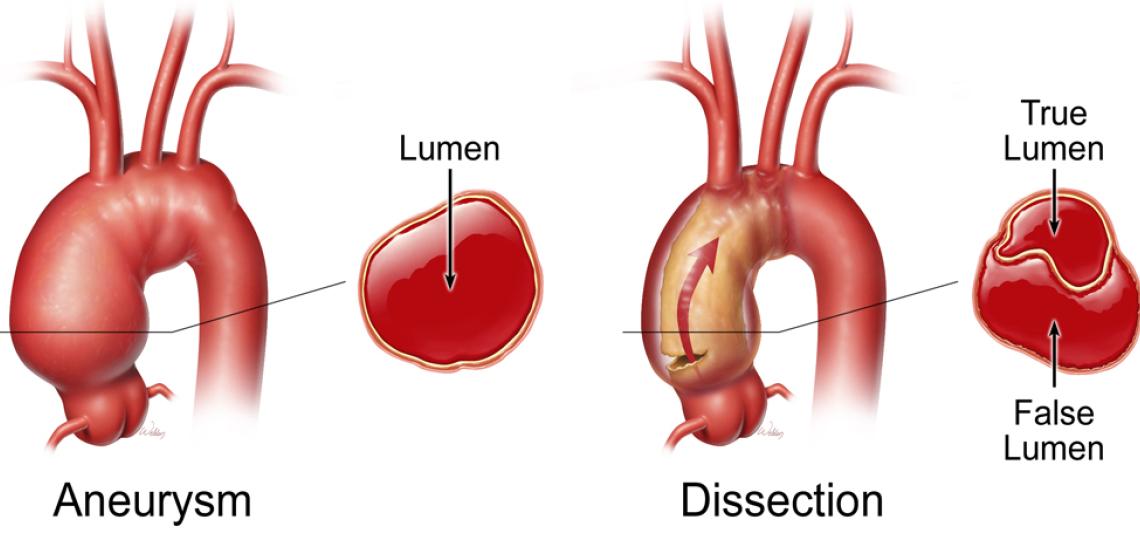
Aortic Disease Research Laboratory
The aortic disease research team pursues several research projects and maintains one of the world’s most extensive and well-cataloged aortic tissue banks. This core resource facilitates investigations into the causes and progression of aortic disease pursued by our researchers, as well as researchers from other academic institutions. The members of the lab employ a wide range of standard and advanced techniques, including tissue analysis, cell-based experiments, mouse models of aortic disease, and single-cell transcriptome analysis.
The lab is working to identify the mechanisms that drive aortic degeneration. Their recent Circulation publication reported that cytosolic DNA and abnormal activation of the cytosolic DNA sensing adaptor stimulator of interferon genes (STING) play a critical role in vascular inflammation and destruction; pharmacological agents inhibiting these genes have been shown to partially prevent aortic aneurysm and dissection. Within the high-volume clinical aortopathy program at Baylor, leading scientists are taking advantage of advances in single-cell analysis technologies to explore the molecular and cellular signatures in the aortic wall that are uniquely associated with the risk factors and conditions of aortic disease.
Laboratory for Cardiac Regeneration
Heart failure remains a leading cause of death and decreased quality of life for millions of patients and traditional treatments have reached the limits of their effectiveness. Researchers in the Laboratory for Cardiac Regeneration, led by Tamer M. Mohamed, Ph.D., M.Sc., are studying whether cellular reprogramming can be applied to improve cardiac remodeling and function after myocardial infarction. By converting scar tissue into new contractile cells, cellular reprogramming can reduce fibrosis and restore cardiac function.
Valve Sparing Approach to Aortic Repair
Under the leadership of Joseph S. Coselli, M.D, the division is active in evaluating the outcomes of both standard and emerging surgical techniques for aortic repair. While the traditional approach toward repair of an aortic root aneurysm involves replacing it with a composite valve graft, an alternate approach—valve-sparing aortic root replacement—has increasingly become more widely used.
New Technologies for Aortic and Valve Repair
Joseph S. Coselli, M.D, also leads the division’s participation in several clinical trials focused on new devices that may improve surgeons’ ability to treat complex aortic and valvular disease.
Aortic Valve Operative Outcomes in Marfan Patients
The Marfan Foundation and Baylor College of Medicine are conducting the multicenter Aortic Valve Operative Outcomes in Marfan Patients (AVOMP) study to compare the outcomes of aortic valve-sparing versus valve-replacing procedures in individuals with Marfan syndrome.
Cardiothoracic Surgical Trials Network
As a Core Clinical Center (CCC) for the NIH/NHLBI-funded Cardiothoracic Surgical Trials Network (CTSN). Led by our chair Todd K. Rosengart, M.D. and Ravi Ghanta, M.D., the Michael E. DeBakey Department of Surgery at Baylor College of Medicine is one of 30 highly experienced cardiothoracic surgical centers participating in the CTSN Tricuspid Repair Trial entitled “Evaluating the Benefit of Concurrent Tricuspid Valve Repair during Mitral Surgery”. In addition, Baylor has been selected as a trial site for the CTSN “Anticoagulation for New-Onset Post-Operative Atrial Fibrillation after CABG (CTSN PACES Trial). Both trials address fundamental questions in the surgical management of ischemic and valvular heart disease, building on the legacy of innovation in cardiovascular surgery at Baylor.