The Agent
HIV (Human Immunodeficiency Virus) causes the disease known as AIDS (Acquired Immune Deficiency Syndrome). HIV infects cells that are critical to the human immune system and destroys their ability to fight infections. Therefore, people who have developed AIDS are unable to fight off infections by other viruses or bacteria that normally do not cause disease in healthy individuals. As a result, AIDS patients can die from infections that are normally harmless. The leading cause of death in people living with HIV/AIDS is tuberculosis. Some AIDS patients also develop unusual cancers because of defects in their immune systems.
HIV was first recognized in 1983 as the agent that causes AIDS, a disease initially found primarily in small clusters of homosexual men. Since that time, around 78 million people have become infected with HIV, and 39 million people have died of AIDS-related illnesses.
In recent years, however, the situation has become more hopeful. More people are living with HIV/AIDS due to an expansion in preventative measures, increased testing, and greater access to antiviral drugs. Overall, the number of new infections and deaths from HIV/AIDS has declined, although progress is not uniform within all geographic areas and population subgroups.
Worldwide HIV/AIDS Epidemiological Statistics
Worldwide, nearly 37 million people were living with HIV in 2014, 70 percent of them in sub-Saharan Africa. However, progress is apparent as the number of new annual infections has fallen by 35 percent since 2000. Furthermore, although the total number of people living with HIV has risen through the years, the number of AIDS-related deaths has dropped by 41 percent, after peaking in 2005. This is a result of vastly increased access to lifesaving antiviral drugs and large global investments in the AIDS response.
As of May 2015, 15 million people living with HIV, or 40 percent of those infected, had access to antiretroviral therapy, a 22-fold increase since 2000, when fewer than 1 percent of HIV-infected people living in low- and middle-income countries had access to treatment. This has resulted in a halt or reversal of new HIV infections in 83 countries, including the hard-hit countries of India, Kenya, South Africa and Zimbabwe.
Success in slowing HIV is especially notable among children. Between 2000 and 2014, the percentage of pregnant women living with HIV who had access to antiretroviral therapy increased to 73 percent, and new infections in children declined by 58 percent. In 2015, Cuba became the first country to eliminate new HIV infections among children. In 2013, an estimated 3.2 million children under the age of 15 were living with HIV.
Although the rate of HIV infections is declining overall, challenges remain in eliminating HIV/AIDS. In Eastern Europe, Central Asia, the Middle East and North Africa, new infections were on the rise between 2000 and 2014. New HIV infections are on the upswing among men who have sex with men, notably in western Europe and North America, where progress had previously been reported. In sub-Saharan Africa, upwards of 70 percent of adults have never been tested for HIV. Cultural and political policies that hinder the prevention and identification of HIV infections remain in place in many countries.
Of the total number of HIV-infected people in 2014, approximately 26 million lived in sub-Saharan Africa. Asia and the Pacific constitute the region with the next largest group of people living with HIV in 2013 (5 million), followed by Latin America (1.7 million). North America and Central Europe had a combined 2.4 million people living with HIV. In the United States, 1.3 million people were living with HIV in 2011, about a quarter of them women.
| 2001 | 2005 | 2014 | |
|---|---|---|---|
| Adults and children living with HIV | 29.8 million | 32.1 million | 36.9 million |
| Adults and children newly infected with HIV | 3.4 million | 2.9 million | 2 million |
| Adult and child deaths due to AIDS | 2..0 million | 2.4 million | 1.2 million |
Source: UNAIDS
Infection
From the time a person becomes infected with HIV, there is usually a substantial lag time, generally about a decade, before an infected person develops the symptoms of AIDS (although this is highly variable from person to person and can be delayed by the use of anti-HIV drugs). Shortly after infection, individuals may experience flu-like symptoms, but then can go for many years without detectable symptoms. However, during this phase, the virus continues to replicate within their bodies and can be transmitted to other people through direct contact with body fluids such as blood or semen.
During this time, there is usually also a slow but steady depletion of the immune system cells known as CD4-positive (CD4+) T cells that are infected by HIV. A normal person has about 1000 CD4+ T cells in a milliliter of blood. Once CD4+ T cell numbers fall to 200 cells per milliliter, the patient enters the phase of HIV infection that is known as AIDS, and it becomes increasingly difficult for these AIDS patients to fight off infections.
The HIV Replication Cycle
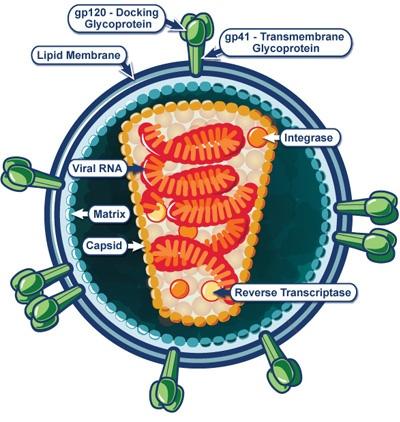
HIV belongs to a class of viruses known as retroviruses. Retroviruses use RNA to encode their genetic information rather than DNA, as human cells do. What distinguishes retroviruses from other classes of viruses is that during their replication cycle inside cells, they use a special enzyme called reverse transcriptase to convert the RNA into DNA.
- To begin an infection, the HIV particle must encounter and bind to the CD4 receptor that is present on the surface of a subset of T cells (and also some other immune system cells called macrophages, but not on most other cells in the body). The interaction between HIV and the CD4 molecule, along with another molecule called a co-receptor, allows HIV to enter the cell.
- The virus enzyme reverse transcriptase then converts the RNA to DNA.
- Another viral enzyme called integrase inserts the virus’ DNA into the cell’s DNA in a step known as integration.
- Next, the cellular machinery, with the help of a virus protein named Tat, makes many RNA copies of the virus in a process called transcription.
- The RNA copies are then used to make virus proteins, and the RNA copies and virus proteins are assembled into new virus particles.
- The long chains of virus proteins in the virus particle have to be cut into smaller pieces by another viral enzyme called protease. The new HIV particles are released from the cell by a process called budding and then can go on to repeat the cycle and infect more and more CD4+ T cells.
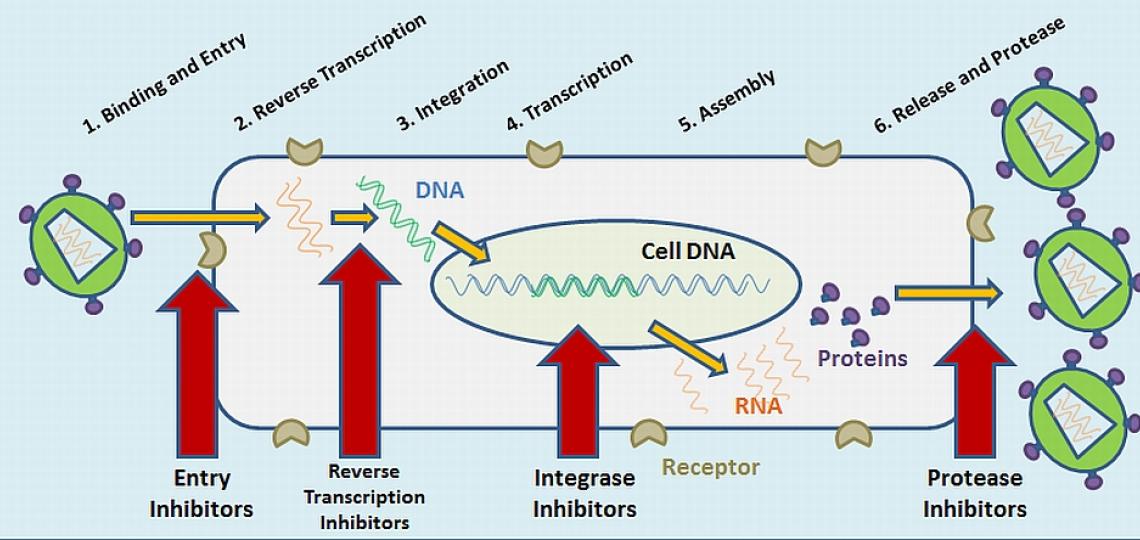
Important steps in the replication of HIV are blocked by anti-HIV drugs.
Anti-HIV Drugs Target Steps in the HIV Replication Cycle
Scientists have spent many years trying to understand the details of the steps in the HIV replication cycle, because these points represent possible targets for drug intervention. Because each of these steps is required for the virus to produce more copies of itself, blocking these steps should prevent the virus from continuing to replicate.
Anti-HIV drugs are available that block several different steps in the HIV replication cycle - the entry of the virus into the cell, the reverse transcription step that converts RNA to DNA, and the protease step, which is needed to produce the right forms of the virus proteins (see diagram). An additional drug was approved in 2007 that inhibits the integrase enzyme that is necessary for the virus to insert itself into the host cell genome.
It is most effective to use a combination of drugs that block multiple steps in the HIV replication cycle, a treatment regimen called HAART (highly active antiretroviral therapy).
The Problem
Antiviral Drugs
There is still no cure for AIDS. Effective therapies against HIV now exist, which extend and improve the quality of life for HIV-infected patients, and reduce transmission of the virus. However, there are a number of problems with the current drugs.
- Their expense limits their accessibility to all people requiring treatment, especially in poorer countries of the world.
- Some patients suffer from unacceptable side effects.
- While the drugs decrease the level of virus in the blood of infected patients, the virus can “hide” in a temporarily inactive or latent form inside cells or in areas of the body that are inaccessible to the drugs.
- When treatment is discontinued, the virus rebounds to high levels.
- The virus can mutate and become resistant to drug treatments.
Vaccine
The ultimate goal is to develop a vaccine that will prevent the transmission of HIV, or at least the progression to AIDS in individuals already infected. However, this remains a great challenge. Clinical trials of candidate vaccines are underway, but the development of effective vaccines has been hampered by:
- The ability of HIV to mutate. Although the immune system may recognize a particular version of HIV used to make a vaccine, it may fail to recognize a mutated version that is present in an infected individual and therefore be unable to prevent infection.
- An incomplete understanding of how the human immune system can best control or eliminate the virus.
Prevention
Until a vaccine or improved drugs can be developed, prevention of new infections is a top priority. Social and political obstacles continue to hinder this goal. Although the means – especially unprotected sexual contact, intravenous drug use, and maternal to infant – by which HIV is transmitted are well established, lack of education and inaccessibility of effective protective measures such as condoms, sterile syringes and maternal prenatal antiretroviral treatment allow the disease to continue to spread.
In 2013, 19 million out of a total of 35 million HIV-infected people were unaware of their HIV-positive status. These individuals have not received treatment and can unknowingly spread the virus to other people.
The Research
There are several researchers who are currently working on the HIV/AIDS problem within the Department of Molecular Virology and Microbiology. The general focus of the work is on understanding the details of how HIV replicates and how it causes disease, and on identifying new potential targets for drug therapies and developing vaccine strategies. The goal is to deepen our knowledge about the virus and its interactions with the host cell and the immune system in order to develop novel antiviral drugs and vaccines.
HIV Cellular Co-factors
Dr. Andrew Rice, who has been conducting HIV research since the mid-1980s, has been studying the transcription part of the virus replication cycle. This is the step in which the virus Tat protein and a large “factory” of cellular proteins make RNA copies of the virus from the virus DNA that is integrated into the cell’s DNA. Several years ago, his group discovered that the virus’ Tat protein does not function alone, but requires two cellular proteins that are called Cdk9 and cyclin T1, which act together to modify the cellular transcription factory and make it much more efficient at making virus RNA copies. These two cellular proteins are therefore essential for HIV to replicate and represent potential drug targets.
Dr. Rice’s group is continuing to study how Cdk9 and Cyclin T1 are regulated in the two major types of immune system cells that HIV infects – CD4+ T cells and macrophages. His laboratory has recently found that a specific phosphorylated form of Cdk9, which is essential for its catalytic function, is highly regulated in CD4+ T cells. This regulation is likely to have profound effects on HIV replication levels. Additionally, his laboratory has identified a microRNA in monocytes that inhibits Cyclin T1 protein expression. This microRNA, therefore, appears to play a role in the repression of HIV replication in monocytes. Increased understanding of how Cdk9 and Cyclin T1 work in cells and the ability to control their activity and interaction with Tat could potentially be used to develop new anti-HIV therapies that would target a step in the virus replication cycle for which there are currently no drugs.
Animal Models of HIV Infection and Disease
MVM researcher Dr. Jason Kimata is interested in studying strategies used by HIV to reproduce and survive in the host with the goal that the results from these studies may help define new ways to prevent HIV infection, inhibit its replication, and provide insight important to the development of a globally effective vaccine. For this purpose, his laboratory is using experimental infection of macaques with simian immunodeficiency virus (SIV) as a model for HIV infection and disease. Like HIV in humans, SIV causes AIDS in Asian macaque species. Dr. Kimata and colleagues have determined that SIV, and by analogy HIV, mutates and becomes more virulent during the course of an infection. They are identifying the changes in the SIV mutants that correlate with increased viral replication and disease-causing potential. They have found mutations that enhance replication of the virus by enabling the virus to evade, exploit, and suppress the immune response of the host. Furthermore, the changes in virulence of SIV may occur because of the host's immune response against the virus. Complementary studies are being conducted with viruses isolated from cohorts of HIV infected individuals.
Dr. Kimata’s lab is also involved in studies to better understand how HIV and SIV are transmitted from one host to another and why some primate species are able to resist infection by HIV and SIV, while others are susceptible to infection and disease. Of note, HIV-1 has a fairly narrow tropism, that is, it is basically limited to infecting humans and great apes, but not macaques. They are working to understand how macaques are able to resist HIV infection, which may lead to novel strategies to prevent HIV transmission.
Using information gained from their studies of HIV transmission, Dr. Kimata and colleagues have been able to devise a method to make HIV-1 replicate in macaques. Thus, they may now have a new animal model of HIV-1 infection and disease. The development of additional animal models for HIV research has been sought by AIDS researchers for many years because current models are not sufficient to study all aspects of HIV infection. Dr. Kimata’s work may provide a new animal model that can be used for studies of novel antiretroviral therapies and vaccines.
Vaccines
Although considerable effort has been made, a vaccine to protect against HIV infection is not yet available. An effective vaccine against sexual transmission of HIV should elicit two types of immune responses – systemic and mucosal. Dr. Cathy Yao is taking a novel approach to develop such a vaccine and to further understand interactions between vaccines and components of the host immune system. Her group is using an immunogen called simian-human virus-like particles (SHIV VLPs) in their HIV vaccine studies. SHIV VLPs are non-infectious virus-like particles that contain HIV proteins but do not contain HIV genetic information. This vaccine can induce both types of immune responses.
Although other groups are working on HIV virus-like particles as vaccines, Dr. Yao’s approach is unique in that:
- It incorporates a protein from the outer surface of the influenza (flu) virus into the HIV virus-like particles so the vaccine can be delivered to a specific site of the respiratory tract to produce a mucosal response.
- It can be modified by incorporating other costimulatory molecules to enhance the binding and activation of the antigen-presenting cells, such as dendritic cells and B cells, so that it can induce potent immune responses.
- It can produce both humoral (antibody) and cellular (cytotoxic T cell) immune responses against HIV.
- It induces immune responses at other mucosal sites, especially the vaginal site, which prevents HIV from sexual transmission in females.
- It activates immune responses without requiring CD4 + T cells – the cells destroyed during HIV infection, and therefore it can be used as a therapeutic vaccine for existing AIDS patients.
The idea is that people will receive the vaccine via an intranasal spray, which will allow the vaccine to target the mucosal layer of a person’s respiratory tract. It is thought that the vaccine may be used in both the prevention of HIV infection and the therapy of those who have already become infected with HIV.
Currently, Dr. Yao and her colleagues are testing how well this vaccine works in small animal models and are trying to understand the molecular mechanisms of the vaccine. They are also testing their modified SHIV VLP vaccines on non-human primates models. Successful results in monkey trials would be followed by vaccine trials for human use.
A phase 2 vaccine trial in humans is underway and being conducted by Dr. Hana El Sahly and colleagues. 2200 HIV-uninfected persons who are at risk for HIV-1 infection through sexual exposure are being enrolled in this multicenter, randomized, controlled, double-blind study. Participants will be injected with a DNA vaccine composed of plasmids encoding four different HIV-1 genes, including variants of the Env glycoprotein from three different clades of HIV. This will be followed by the injection of multiple HIV genes from different clades that will be introduced using an engineered adenovirus vector as a means to boost the initial immune response. Half of the study participants will be injected with a placebo in place of the HIV genes. The participants will be monitored for up to five years. The goal of this study is to determine the safety of the vaccine and to evaluate the ability of the vaccine to reduce HIV transmission or the viral load, relative to the placebo, in participants who subsequently become infected with HIV.
Center for AIDS Research
In addition to the HIV projects headed by individual researchers, one of a nationwide network of AIDS Research Centers, the Baylor-UTHouston Center for AIDS Research was based within MVM from 1994 until 2016. The CFAR is a federally funded program that facilitates research aimed at understanding, detecting, preventing, and treating HIV/AIDS. The Baylor-UTHouston CFAR was comprised of over 100 members located at BCM, UT-Houston Health Science Center, and other institutions within the Texas Medical Center. It was directed by Dr. Janet Butel, former chair of MVM and a Distinguished Service Professor. Particular strengths of the Baylor-UTHouston CFAR included pediatric AIDS clinical studies, basic virology, and education programs. The Baylor International Pediatric AIDS Initiative, headed by Dr. Mark Kline, a CFAR member, established high-quality care centers that continue to provide services for pediatric AIDS patients in a number of countries, including Romania, Botswana, Lesotho, Swaziland and Uganda.
Learn more about some of the technical terms found on this page by visiting our glossary of terms.