Indications For Use
ER belongs to a superfamily of nuclear hormone receptors. It functions as a transcription factor, when activated by its ligand estrogen, and plays a key role in regulating growth, differentiation, and tumorigenesis in breast. There are two known isoforms of estrogen receptor: ERα and ERβ. The current assays in clinical breast cancer measure only ERα. Nearly all the laboratories today measure ER by immunohistochemistry.
ER is expressed in about 70 percent of invasive breast cancers (1, 2). It is a weak prognostic factor but a strong predictive factor for response to endocrine therapies, both in adjuvant and metastatic settings (3). The primary indication to assess ER in breast cancer is to predict response to hormonal therapies such as tamoxifen, other selective estrogen receptor modulators (SERMs), aromatase inhibitors, etc. The College of American Pathologists (CAP) and American Society of Clinical Oncology (ASCO) have recommended that ER should be measured in all primary breast cancers using validated biochemical or immunohistochemical methods (4, 5). ER has also recently been shown to predict for benefit from tamoxifen in patients with ductal carcinoma in situ (DCIS) (6).
Scoring/Interpretation
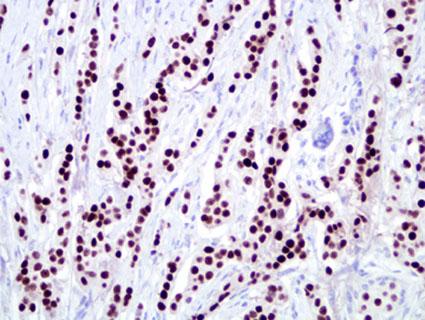
ER immunostaining is scored in our laboratory using the "Allred Score" which is a microscopic method conveying the estimated proportion and intensity of positive tumor cells (range from 0-8). This scoring system was developed in our laboratory over a decade ago and has been used and validated for several markers, including ER, PR, HER-2/neu and p53. Based on several large clinical studies using this scoring system and an assay based on antibody 6F11 (specific for ERα), our laboratory has determined that "3" (corresponding to 1-10 percent positive cells) the optimum score for defining ER-positive breast cancer (7-9). See detailed staining procedure.
Selected Bibliography
Elledge RM, Allred DC. Clinical aspects of estrogen and progesterone receptors. In: Harris JR, Lippman ME, Morrow M, Osborne CK, editors. Diseases of the breast. Philadelphia: Lippincott Williams & Wilkins; 2004. p. 603-17.
Allred DC, Harvey JM, Berardo M, Clark GM. Prognostic and predictive factors in breast cancer by immunohistochemical analysis. Mod Pathol 1998;11(2):155-68.
Tamoxifen for early breast breast cancer: an overview of the randomised trials. Early Breast Cancer Trialists' Collaborative Group. Lancet 1998;351:1451-67.
1997 update of recommendations for the use of tumor markers in breast and colorectal cancer. Adopted on Nov. 7, 1997, by the American Society of Clinical Oncology. J Clin Oncol 1998;16(2):793-5.
Fitzgibbons PL, Page DL, Weaver D, Thor AD, Allred DC, Clark GM, et al. Prognostic factors in breast cancer. College of American Pathologists Consensus Statement 1999. Arch Pathol Lab Med 2000;124(7):966-78.
Allred DC, Bryant J, Land S, Paik S, Fisher ER, Julian T, et al. Estrogen receptor expression as a predictive marker of the effectiveness of tamoxifen in the treatment of DCIS: Findings from NSABP Protocol B-24. Breast Cancer Res Treat 2002;76(1):S36 (abstr #30).
Harvey JM, Clark GM, Osborne CK, Allred DC. Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancer. J Clin Oncol 1999;17(5):1474-81.
Elledge RM, Green S, Pugh R, Allred DC, Clark GM, Hill J, et al. Estrogen receptor (ER) and progesterone receptor (PgR), by ligand-binding assay compared with ER, PgR and pS2, by immuno-histochemistry in predicting response to tamoxifen in metastatic breast cancer: a Southwest Oncology Group Study. Int J Cancer 2000;89(2):111-7.
Love RR, Duc NB, Allred DC, Binh NC, Dinh NV, Kha NN, et al. Oophorectomy and tamoxifen adjuvant therapy in premenopausal Vietnamese and Chinese women with operable breast cancer. J Clin Oncol 2002;20(10):2559-66.








