
About the Lab
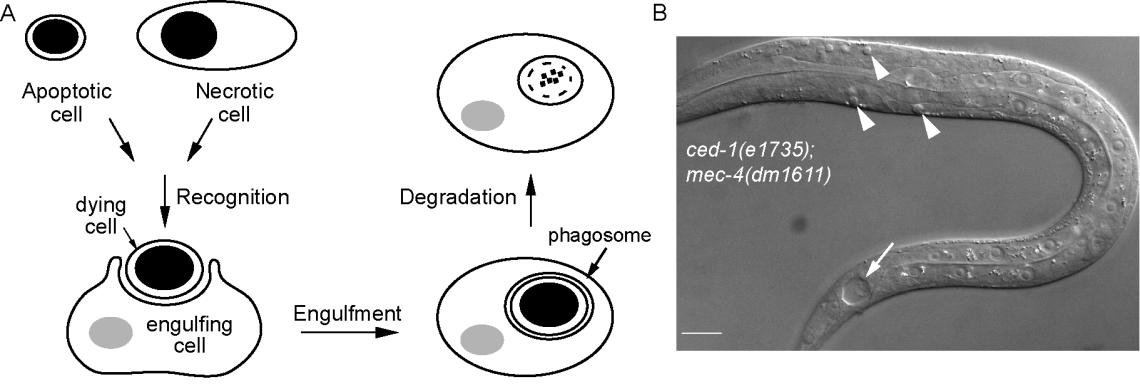
The Zhou Lab is interested in understanding how cells that die inside animal bodies are cleared. During an animal's development and adulthood many unwanted cells are eliminated by a process called "programmed cell death" or "apoptosis". Such cells undergo specific changes in appearance, die, and are quickly engulfed and digested by phagocytes, or engulfing cells. In addition, cells die due to injury, disease, or other pathological states, the “necrotic cells”, are also phagocytosed and degraded efficiently within animal bodies. The clearance of dying cells is important because dying cells contain material that, if released, could harm neighboring cells. Inefficient removal of dying cells results in human autoimmune and inflammatory diseases.
On the other hand, targeted phagocytosis of cancer cells has recently been developed as a cancer treatment strategy. Understanding the mechanisms that control the recognition, engulfment, and degradation of dying cells thus sheds lights on many important issues in the biological and medical researches. However, despite the burst in the study of cell death mechanisms, the molecular mechanisms behind the removal of dying cells remain less known.
We use the free-living soil nematode the Caenorhabditis elegans as a model organism to identify genes and delineate the pathways controlling these events, with the belief that what we learn from C. elegans will be translated to humans. We use primarily genetic, molecular, and cell biological tools for our research. Real-time microscopic imaging at high resolution is an essential approach we employ to observe the dynamic events of protein molecules and subcellular organelles during the clearance of dying cells inside C. elegans embryos. Forward genetic screen and RNA-interference (RNAi) screen allow us to identify mutants defective in specific aspects of the recognition, engulfment, and degradation steps. Genetic and molecular interaction studies enable us to build pathways that drive each event for the clearance of dying cells.

Meet our Team
View a listing of the Zhou Lab team members along with links to their bios.

Our Research
View questions and answers related to how cells that die inside animal bodies are cleared.

Lab News
The Zhou Lab's research was featured in From the Labs: In a cell-eat-cell world calcium ions regulate exposure of ‘eat-me’ signal in necrotic cells.