
Discovery in the Lab
Dr. Rasband's team is discovering the functional organization of axons in health as well as disease or injury.

Our Researchers
Dr. Hamdan at the microscope.
Subcellular Localization of Ion Channels in Neurons
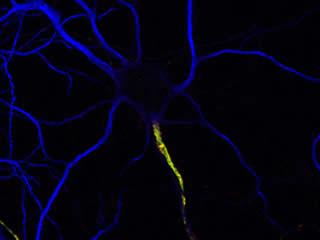
The generation, propagation, and modulation of electrical signals in the nervous system require the precise subcellular localization of ion channels in neurons. For example, disrupted ion channel or neurotransmitter receptor distribution or density play important roles in neuropathic pain, spinal cord injury, schizophrenia, myasthenia gravis, epilepsy, and multiple sclerosis. The three best examples of ion channel/receptor clustering in the nervous system are found at the synapse, the node of Ranvier, and the axon initial segment (AIS).
In the case of the synapse, the identification and characterization of synaptic protein components and complexes has led to remarkable advances in our understanding of synaptic physiology and function. We are even beginning to understand some of the rules that govern synaptic protein localization and retention. In contrast to the synapse and despite their central role in initiating, modulating, and transmitting electrical signals throughout the nervous system, very little is known about the mechanisms regulating localization and retention of proteins at the AIS and nodes of Ranvier.
Furthermore, very little is known about the consequences of nervous system injury on the maintenance and re-assembly of these critical domains.
Nodes of Ranvier
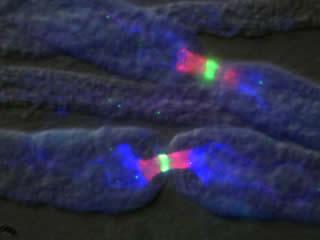
Intriguingly, nearly every identified neuronal protein component of nodes of Ranvier is also found at the AIS, suggesting that these domains perform similar tasks and that common mechanisms may underlie their formation and maintenance. On the other hand, despite the molecular similarities between these two neuronal domains, there is one major difference between the AIS and node of Ranvier: clustering of proteins at nodes requires the influence of myelinating glia, but recruitment of these same proteins to the AIS does not.
Recent work from several laboratories, including my own, have now shown that the AIS is intrinsically specified by the neuron, but node of Ranvier formation and placement is dictated by extrinsic, glial derived factors. The Rasband laboratory is working to elucidate the molecular mechanisms that are responsible for assembly and maintenance of electrogenic compartments of axons in normal health, and during disease or injury. Any therapeutic strategy aimed at nervous system repair, regeneration, or protection, will require the proper assembly and/or maintenance of axon initial segments and nodes of Ranvier.