Testing tuberculosis vaccine for COVID-19
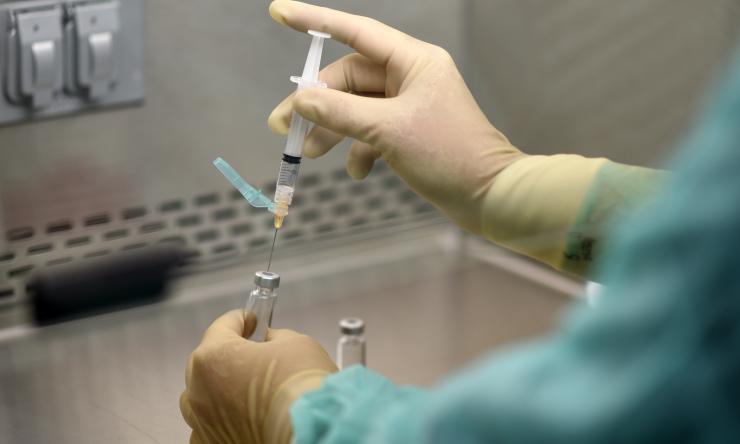
A potential tool in the fight against COVID-19 could be found in a vaccine that already is being used widely around the world. The BCG, or bacille Calmette-Guerin, vaccine prevents tuberculosis and is one of the oldest vaccines still in clinical use. Researchers at Baylor College of Medicine and four other institutions around the country are working to find out if it also can work against COVID-19. They are now recruiting for a clinical trial to test the vaccine.
“Epidemiological studies show that if you’re BCG vaccinated, you have a decreased rate of other infections,” said Dr. Andrew DiNardo, assistant professor of medicine – infectious diseases at Baylor.
The BCG vaccine has proven to help with an immune response to yellow fever and influenza. DiNardo said the vaccine could show 30 to 50 percent improvement in immune response against a pathogen like SARS-CoV-2.
In addition to epidemiological evidence of BCG’s impact on immune response, DiNardo said there is molecular evidence that BCG changes the way the body responds to a pathogen. The vaccine has been shown to rearrange DNA organization so different genes can be expressed.
“Think of DNA like a ball of yarn. Some pieces of the ball of yarn are open and able to be expressed,” DiNardo said. “Other pieces are wrapped up tight and hidden away, and those genes are repressed. It’s a normal way for cells to turn certain genes on and off. BCG opens up certain parts of this ball of yarn and allows the immune system to act quicker.”
Testing the vaccine in a clinical trial
Baylor researchers are testing this immune response in a clinical trial involving healthcare workers who treat COVID-19 patients and are most likely to be exposed to the virus. They are partnering with multidisciplinary researchers at Texas A&M University, the University of Texas MD Anderson Cancer Center, Cedars-Sinai Medical Center in Los Angeles and Harvard University.
Participants at Baylor will receive either the BCG vaccine or a placebo. They will report progress through a questionnaire on a smartphone every few days for six months. DiNardo said this vaccine should not be considered a definitive prevention for COVID-19. But he is cautiously optimistic that this vaccine could work as a stop-gap measure.
“It is a very well-known and safe vaccine available now that might be able to work as a buffer until a COVID-19 vaccine is developed, which could take 18 months,” DiNardo said. “When a COVID-19 vaccine comes out, the BCG vaccine should be tested with it to see if it improves the efficacy.”
Vaccine already in trials for bladder cancer
The BCG vaccine is widely used to prevent tuberculosis around the world, except in the U.S. and Western Europe where TB infection is not common. BCG also is used to treat patients with high-grade, non-muscle invasive bladder cancer by instilling it inside the bladder via a catheter. This is a standard of care for urologists throughout the world and the most effective therapy for these patients. Dr. Seth Lerner, professor of urology and the Beth and Dave Swaim Chair in Urologic Oncology at Baylor, is overseeing a National Cancer Institute funded clinical trial led by the SWOG Cooperative Group. The trial is comparing two BCG strains for bladder cancer treatment and adding vaccination with BCG to see if this will help boost the immune response to BCG and improve outcomes for the bladder cancer patients (NCT 03091660). He is lending his expertise to the COVID-19 clinical trial.
“There are some signs that suggest a more systemic response to the bladder treatment,” Lerner said. “Some patients are asking if it will help protect against COVID-19, and we don’t know the answer. But hopefully with this new clinical trial, we will find out.”
People interested in participating can send an email to dinardo@bcm.edu and sharons@bcm.edu.