Proteomic determinants of response to checkpoint blockade in malignant pleural mesothelioma
(R37CA248478): We aim to investigate the immunoproteogenomic determinants that predict responses to checkpoint blockades in pleural mesothelioma.
Single-cell multi-omics integration to exploit autoimmunity induced by checkpoint immunotherapy in malignant pleural mesothelioma
(Department of Defense): We aim to define autoimmune susceptibility and elucidate novel cellular and molecular mechanisms underlying immunotoxicity following immune checkpoint therapy in pleural mesothelioma.
Novel humoral and cellular biomarkers of autoimmune diseases caused by immunotherapy
(R21AI159379): We aim to investigate the immunoproteogenomic determinants that predict responses to checkpoint blockades in pleural mesothelioma.
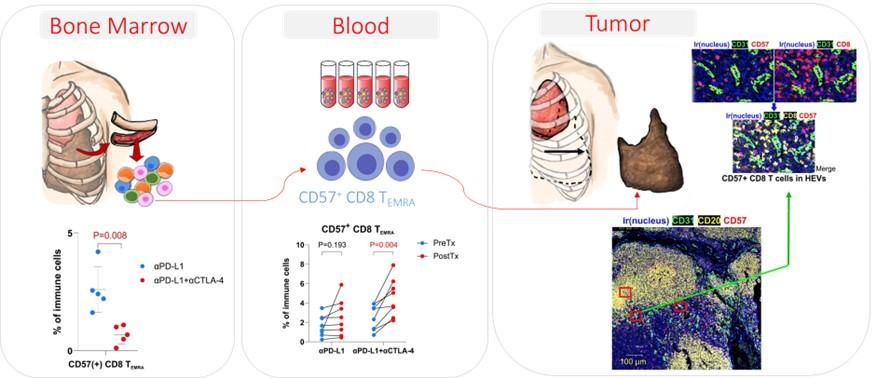
Unraveling NK-like CD8 T cell dynamics for advancing precision immunotherapy
(Helis research grant): We aim to elucidate the mechanisms of immune-related adverse events (irAEs) by investigating the intersection between anti-tumor immunity and autoimmunity, and to develop preclinical therapeutic strategies for their effective management.
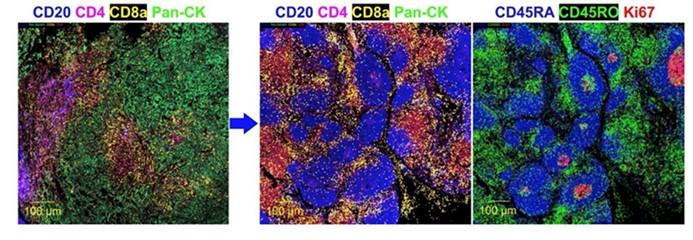
Spatial Profiling of Tumor-Immune Microenvironment by Multiplexed Single- Cell Imaging Mass Cytometry
(CPRIT RP200443): We aim to create spatial biomarkers that determine which tumors can be effectively treated with immune checkpoint inhibitors and to identify novel therapeutic approach to overcome resistance to checkpoint immunotherapy.
Therapeutic targeting of MIC-NKG2D signal to overcome resistance to checkpoint immunotherapy
(Samyang Biopharmaceutical USA): We aim to investigate and study the role of anti-MIC (MHC Class I Chain-Related Protein) antibody in overcoming resistance to checkpoint immunotherapy by targeting MIC-NKG2D (a receptor on natural killer cells and cytotoxic T cells) through systems onco-immunology approach. This investigation will lay a solid foundation for future clinical trials.








