Background and Significance: Advancing Human Disease Genetics Using Drosophila Models

Genetic studies in model organisms, including Drosophila, have a rich history of fundamental insights in neuroscience, including the identification and functional dissection of genes important in human disease. The fly nervous system operates on the same fundamental biological principles as its human counterpart, and many neurotransmitter systems including dopamine, acetylcholine, glutamate, and GABA are conserved from flies to humans. In addition, the Drosophila and human genomes share substantial homology, and the majority of human disease genes are conserved in flies. Compared to mammalian model systems,Drosophila offers the power of rapid and high-throughput genetic analysis, with a generation time ten times faster than the mouse. Further, fly models have now been developed for investigating a variety of neurodegenerative diseases, and these systems have demonstrated their utility for genetic dissection of disease mechanisms (Shulman et al. 2003 ). For example, when expressed in the Drosophila brain, Tau protein—which forms neurofibrillary tangle pathology in AD—causes adult onset, progressive neurodegeneration, truncated lifespan and increased phosphorylation and aggregation of Tau, thereby recapitulating several key features of human disease (Wittman et al. 2001).
We have participated in a genetic screen to identify enhancers and suppressors of Tau neurotoxicity in flies, revealing a number of molecular pathways with roles in AD pathogenesis (Shulman and Feany 2003). More recently, we have developed and validated a strategy that leverages Drosophila genetics to enhance the power of human genetic studies (Shulman et al. 2011). Despite the promise of genome-wide association studies, discovered polymorphisms rarely definitively identify causal susceptibility genes, but instead highlight broad genomic regions. In a pilot study, we used the Tau transgenic flies for functional validation of results from a genome-wide scan for AD neuropathologic burden. In 6 out of 15 genomic regions, we successfully identified a candidate causal gene for the association, based on in vivo genetic interactions with Tau neurotoxicity. We have extended this strategy to consider 159 independent loci achieving significant (p<5x10-8) or suggestive (p<10-4) associations with AD from GWAS. Ten Drosophila genes, orthologous to candidate human AD susceptibility genes, were validated in our functional screen. For example, loss-of-function in cindr, the fly ortholog of human CD2AP, enhances both Tau-induced retinal toxicity and age-dependent neurodegeneration in the brain (Shulman et al., unpublished data). CD2AP/cindr is a regulator of actin dynamics, and several other validated genes further implicate cell-adhesion pathways converging on the cytoskeleton as important in Tau neurotoxicity and AD susceptibility.
Current Projects
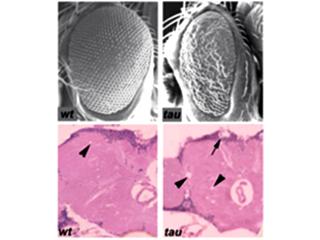
Building on our proven approach, we are significantly increasing the scope of functional screening in Drosophila disease models in order to accelerate the validation of human susceptibility genes and elucidation of their mechanisms of action. We continue to evaluate fly orthologs of emerging AD susceptibility genes, and the number of loci is projected to sharply increase with the completion of the international AD GWAS meta-analysis, incipient sequencing efforts to identify rare variants, and from our own analyses focused on AD pathology endophenotypes. In addition to Tau transgenic flies, we will perform functional studies based on expression of the human amyloid-Beta peptide (Aß), which is similarly associated with neuronal toxicity in Drosophila. Besides the "rough eye" phenotype generated by Tau and Aß retinal degeneration, we are developing additional assays that support a high-throughput and in vivo genetic validation strategy, allowing us to comprehensively evaluate promising loci from human genomic studies. RNA-interference (RNAi) lines are available for nearly all fly genes, as well as putative overexpression lines for a substantial fraction. Positive "hits" identified as modifiers of Tau and/or Aß toxicity are secondarily tested for modulation of aging-dependent neurodegeneration in the fly brain, and we employ a variety of scalable assays for efficient dissection of susceptibility gene mechanisms, including potential roles in protein aggregation, phosphorylation, oxidative damage, ER stress, cytoskeletal changes, or apoptosis.
Cellular networks implicated in AD susceptibility are being investigated in greater depth, using the full power and flexibility of Drosophila genetic methods. For example, we are currently following up our finding that cell adhesion pathways are important for Tau-mediated neurotoxicity. Detailed mapping of these pathways and the associated molecular mechanisms will also generate hypotheses for reciprocal genetic studies in human population samples. This integrative, cross-species approach is a powerful strategy for functional genomic dissection of neurodegenerative disease.
A major goal of the laboratory is to develop an analogous experimental approach for enhancing genetic investigation of Parkinson's disease (PD). Expression of human alpha-synuclein in the fly nervous system is associated with adult-onset dopaminergic cell loss, Lewy body-like inclusions, and disrupted motor behavior. It is therefore an ideal model system for functional validation of PD susceptibility loci for interactions with alpha-synuclein aggregation and neurotoxicity in vivo. We will initially focus on genes identified by PD GWAS consortia and sequencing studies, and we also plan to follow-up novel loci from our studies of PD endophenotypes.