Role of Neurofibromin in the Bone Mesenchymal Lineage
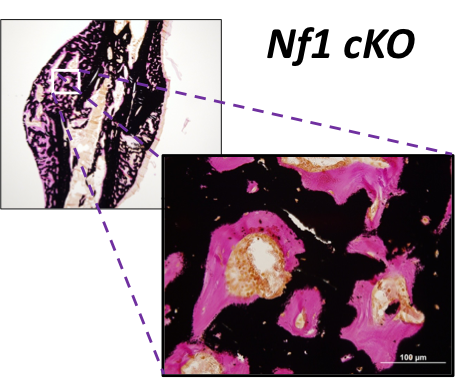
By studying how Nf1 loss-of-function impacts the maturation and function of chondrocytes and osteoblasts, we seek to understand the etiology of the most problematic skeletal maladies associated with neurofibromatosis type 1 (NF1), which includes dystrophic scoliosis, tibia bowing and recalcitrant bone healing.
Growth Plate and Intervertebral Disc Development
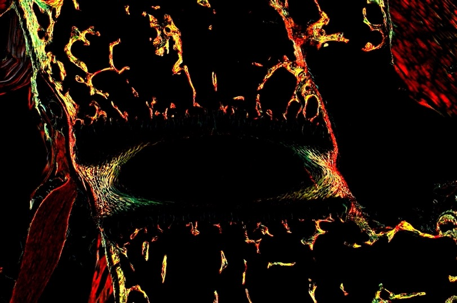
We are interested in better understanding how cells in growth plates and intervertebral discs can actively proliferate and generate their rich extracellular matrix, while under hypoxia, low nutrient availability and hyperosmotic conditions.
Influence of Autonomic Nerves on Bone Remodeling and Metastasis
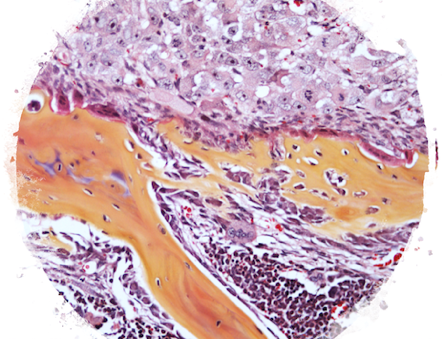
In this project, we seek to better understand how autonomic nerves control the process of bone remodeling, and to determine the pathophysiological relevance of this interaction. Studies are ongoing to determine if the continued bone loss following acquisition of peak bone mass can be explained by increased activity of sympathetic nerves and/or by the reuptake of norepinephrine into the large osteocytic network, and how these nerves favor the skeletal establishment and growth of bone metastatic cancer cells.








