Project Co-leaders
- Eric Chang, Ph.D. (Basic Science Leader)
- Bora Lim, M.D. (Clinical Science Leader)
Project Description
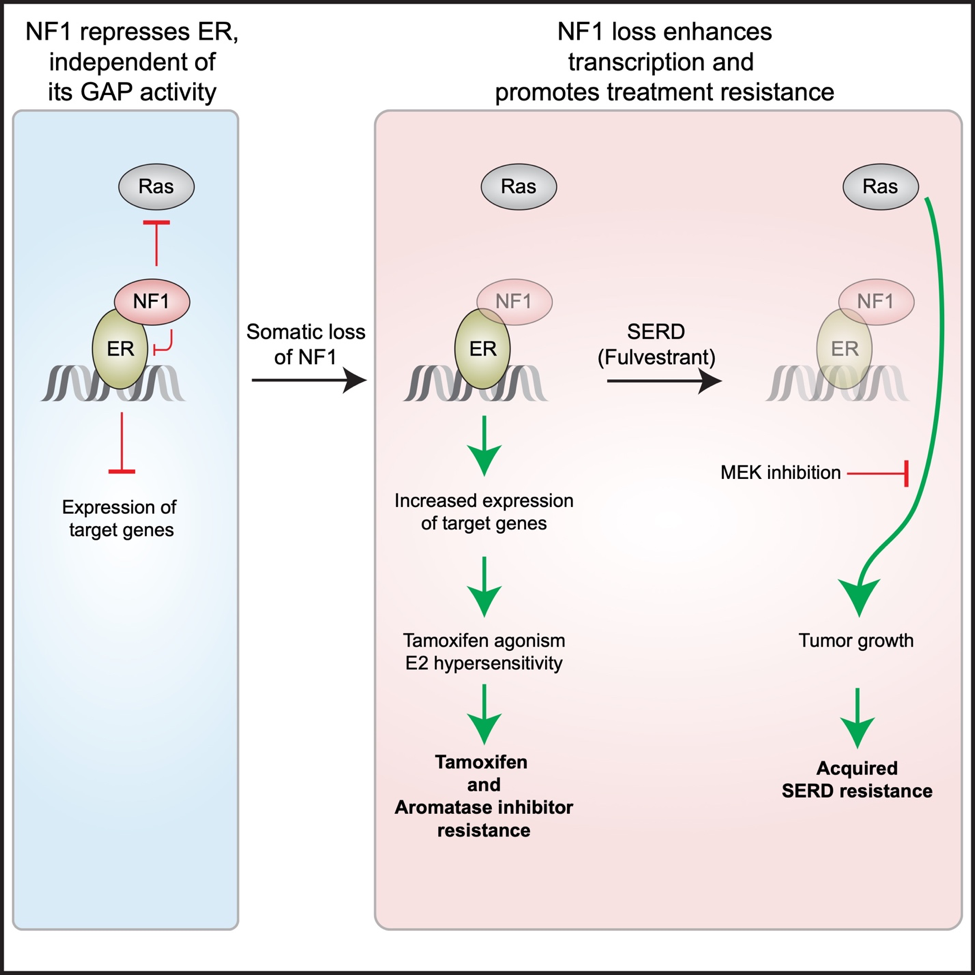
Over 250,000 cases of breast cancer are diagnosed annually in the United States alone, and about 80% of these will be estrogen receptor-α positive (ER+). Despite great progress in treating ER+ breast cancer with endocrine therapy, treatment resistance remains a major problem, causing majority of deaths overall. Our clinical studies on endocrine therapy resistance have found that inactivation of the NF1 (for neurofibromatosis type 1) gene, which may occur in as many as 20% of primary ER+ breast cancer patients, correlates with poor outcome. NF1 encodes neurofibromin, best known as a Ras repressor (a GTPase Activating Protein, or GAP), but the scientific premise of this project stems from our discovery that NF1 has a GAP-independent activity by also acting as an ER transcriptional co-repressor.
When the NF1-ER interaction is selectively inhibited (without affecting GAP activity), ER is de-repressed, estradiol (E2)-dependent gene expression is enhanced, and ER+ cells can grow in low E2 levels observed in aromatase inhibitor treated patients and are stimulated by tamoxifen, not inhibited. In contrast, NF1-depleted cells still respond to fulvestrant, a SERD (Selective ER Degrader), and although the reduced repression of Ras-Raf signaling in NF1-depleted cells promotes acquired resistance, which can be blocked by an FDA-approved MEK inhibitor (MEKi). This project will therefore investigate the hypothesis that NF1-deficient ER+ breast tumors should be treated by a SERD together with inhibitors blocking the Ras-Raf pathway.
The key scientific premise is published in a Cancer Cell paper (Zheng et al 2020) and its graphic abstract is presented below: