About the Lab
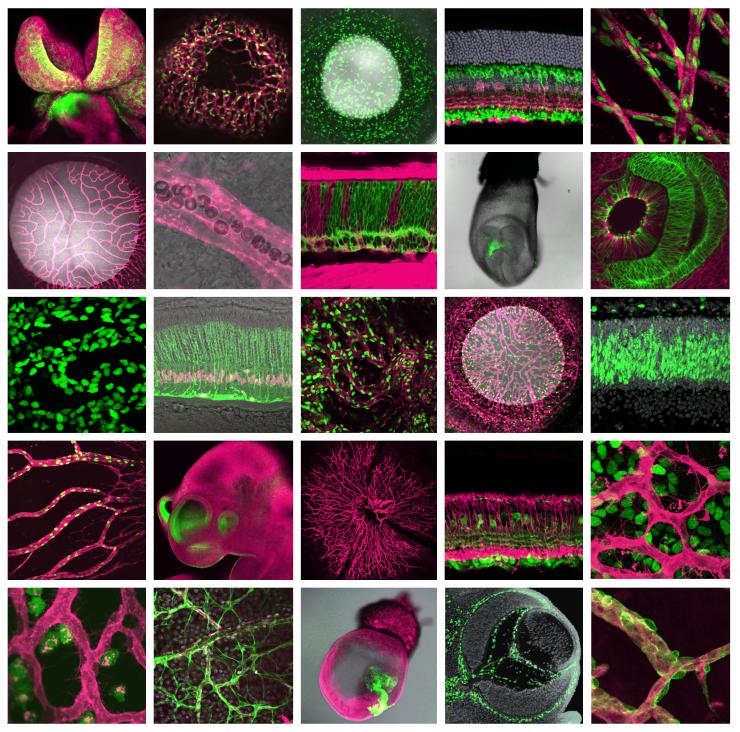
One of my lab's long-term goals is to elucidate the transcriptional and epigenetic mechanisms regulating retinal progenitor cell proliferation and differentiation, leading to new therapeutic interventions to restore sight and treat cancers such as retinoblastoma. Toward that end, projects aimed at characterizing mouse mutants suffering from defects in retinogenesis are ongoing. One specific aspect of our research is to determine precisely how mitochondrial activity and other bioenergetic pathways interface with cell cycle progression during development. Additionally, we aim to identify new strategies to promote retinal regeneration in response to photoreceptor damage. Here, we are interested in determining whether the mouse retina retains latent regenerative potential akin to other vertebrates such as the zebrafish and whether we can genetically "awaken" that potential to restore sight.
Our second focus is on the generation and characterization of novel mouse models recapitulating human craniofacial and neurodevelopmental birth defects. Recently, we have uncovered a previously unknown transcription factor network that is responsible for development of the neural crest-derived craniofacial skeleton.
In the lab, we employ (and enjoy) a multi-disciplinary approach utilizing genetic loss- and gain-of-function experiments, molecular biology and live retinal confocal microscopy.
Recent Lab News
Drs. Annita Achilleos and Elda Reuda have received tenure-track faculty jobs! Annita will be assistant professor, University of Nicosia Medical School in Nicosia, Cyprus. Elda will be assistant professor, University of Houston – Downtown.
In the News
Dr. Ross Poché and research colleagues uncover mechanism blocking retina regeneration. This discovery opens the possibility of one day restoring loss of vision by activating the retina’s ability to regenerate.

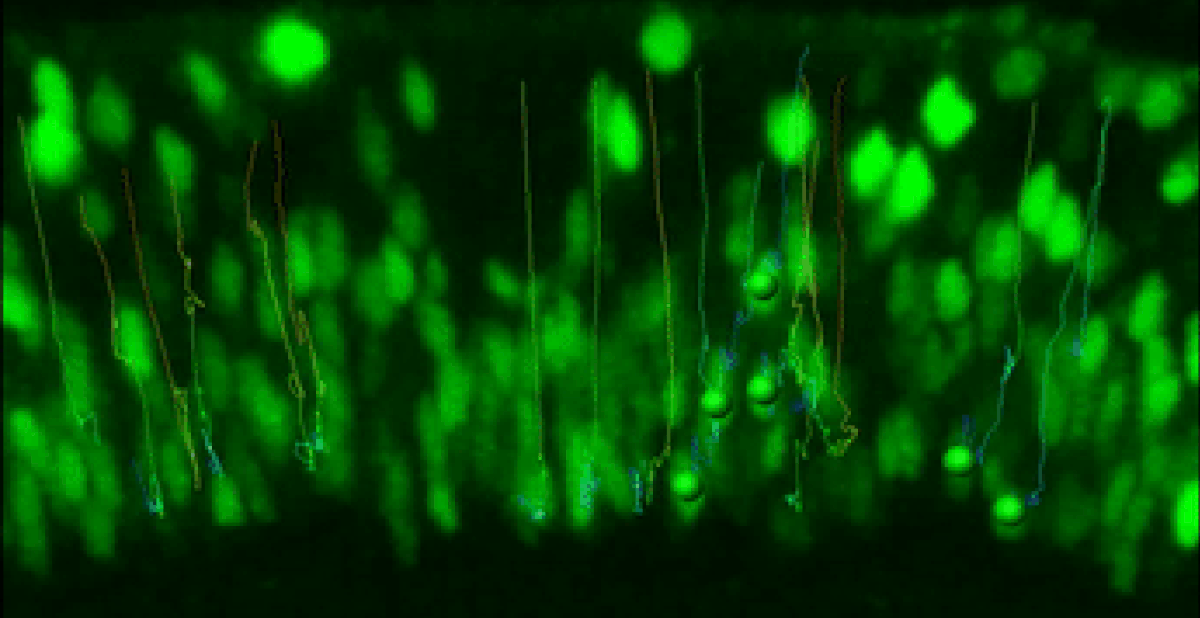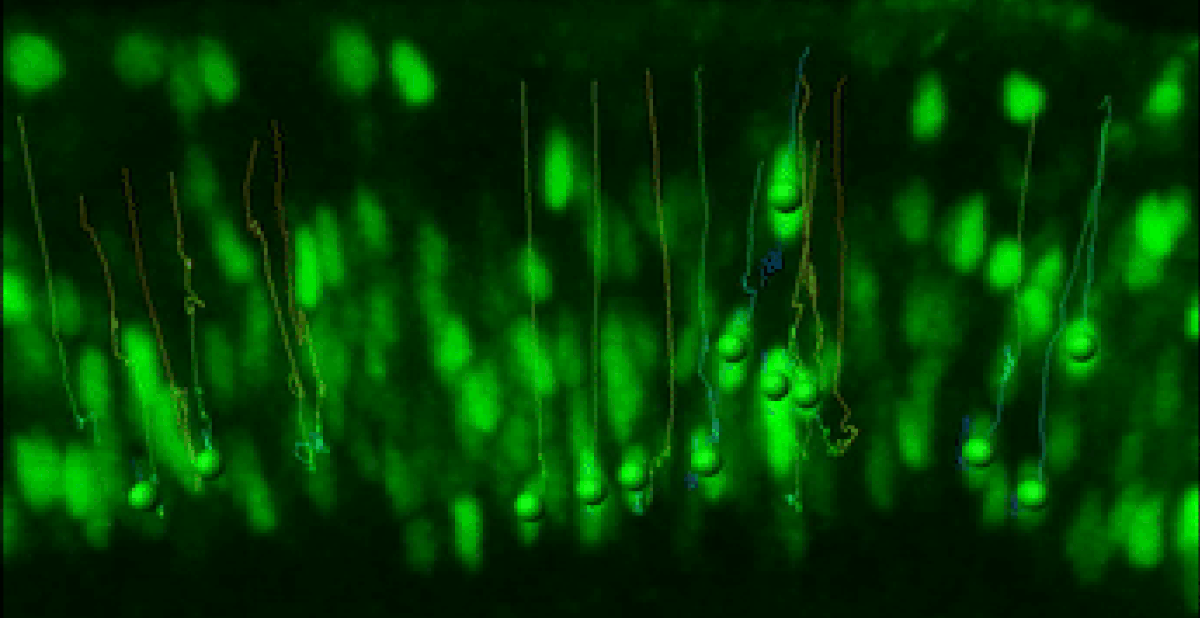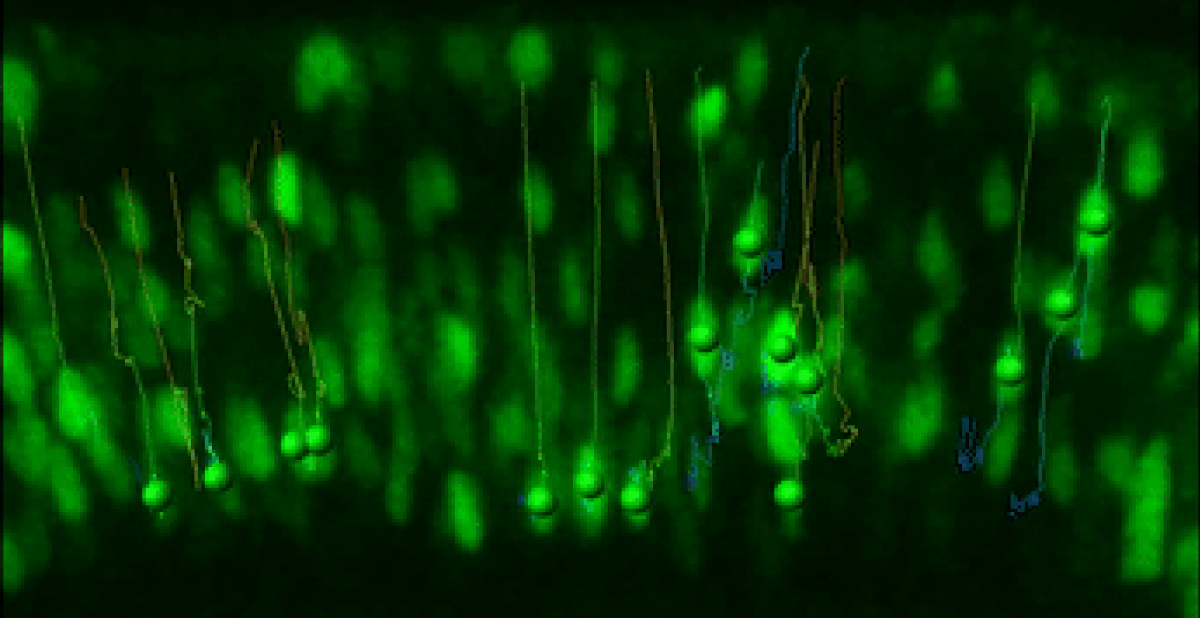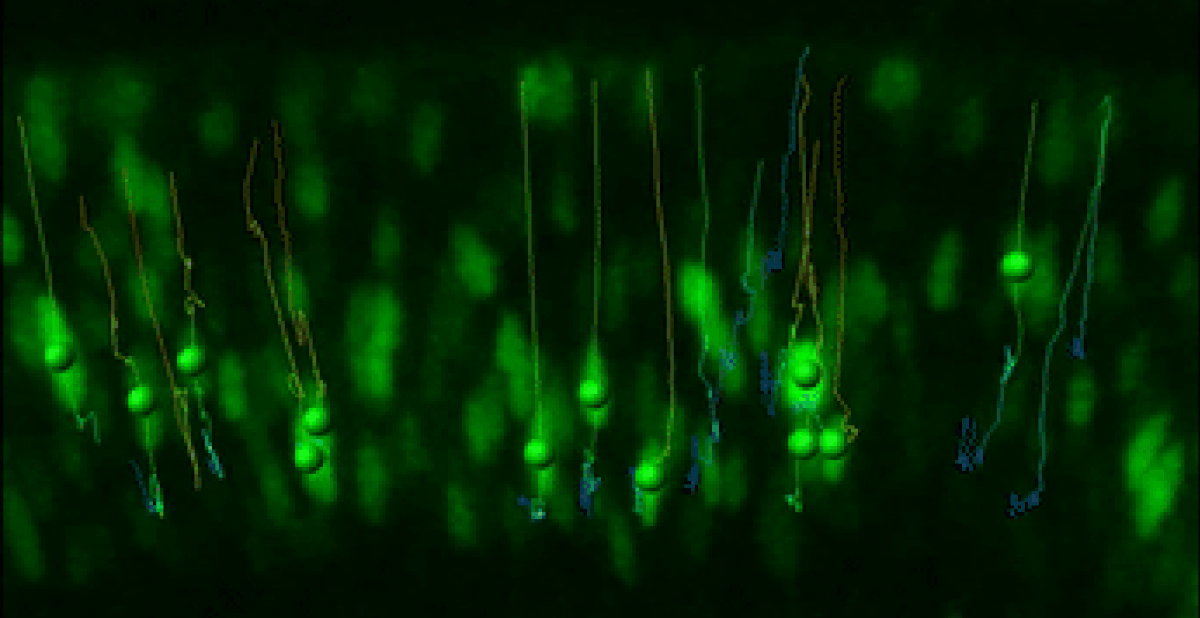
Live Retinal Imaging
We have developed a robust live imaging method to monitor retinal progenitor cell interkinetic nuclear migration coincident with cell cycle kinetics.
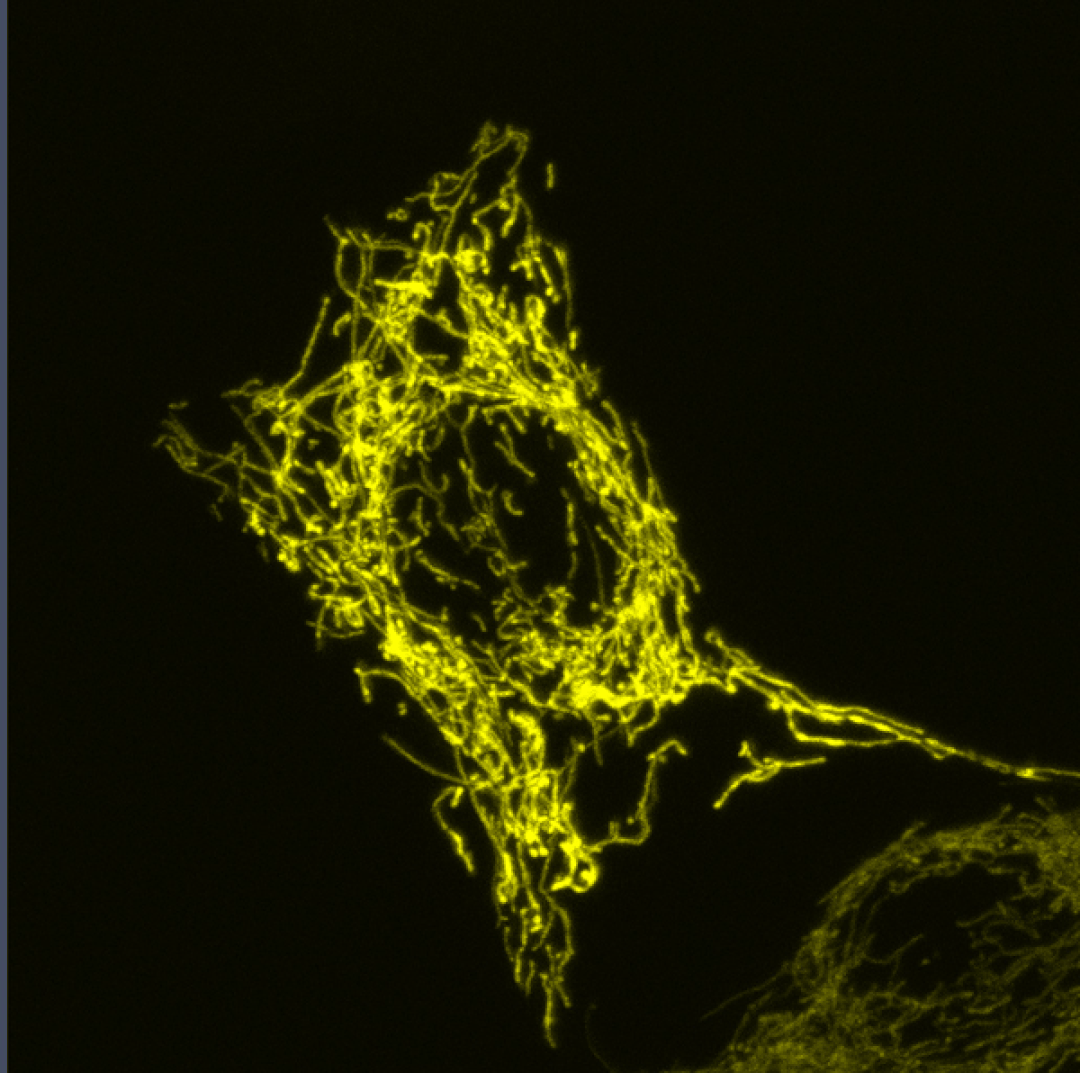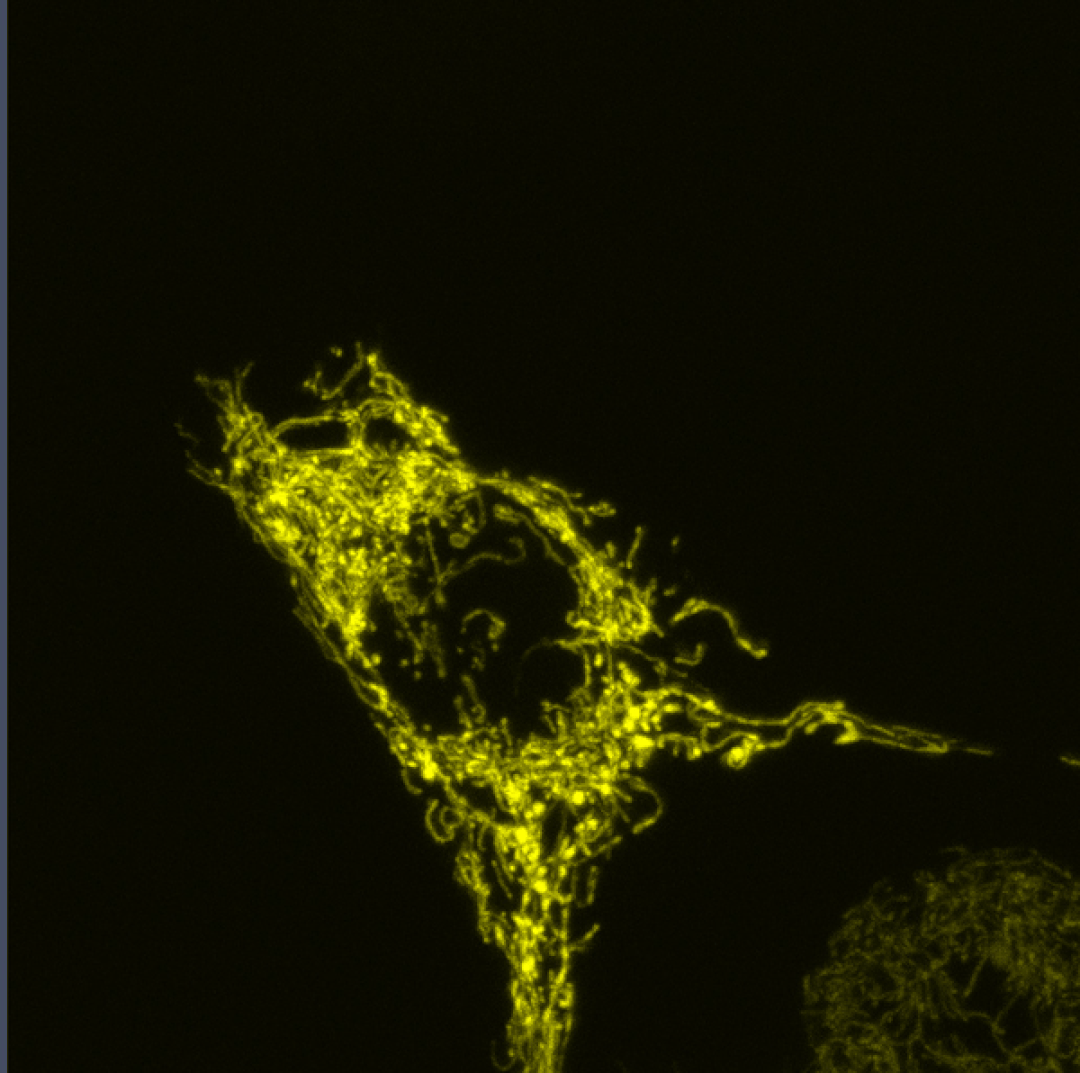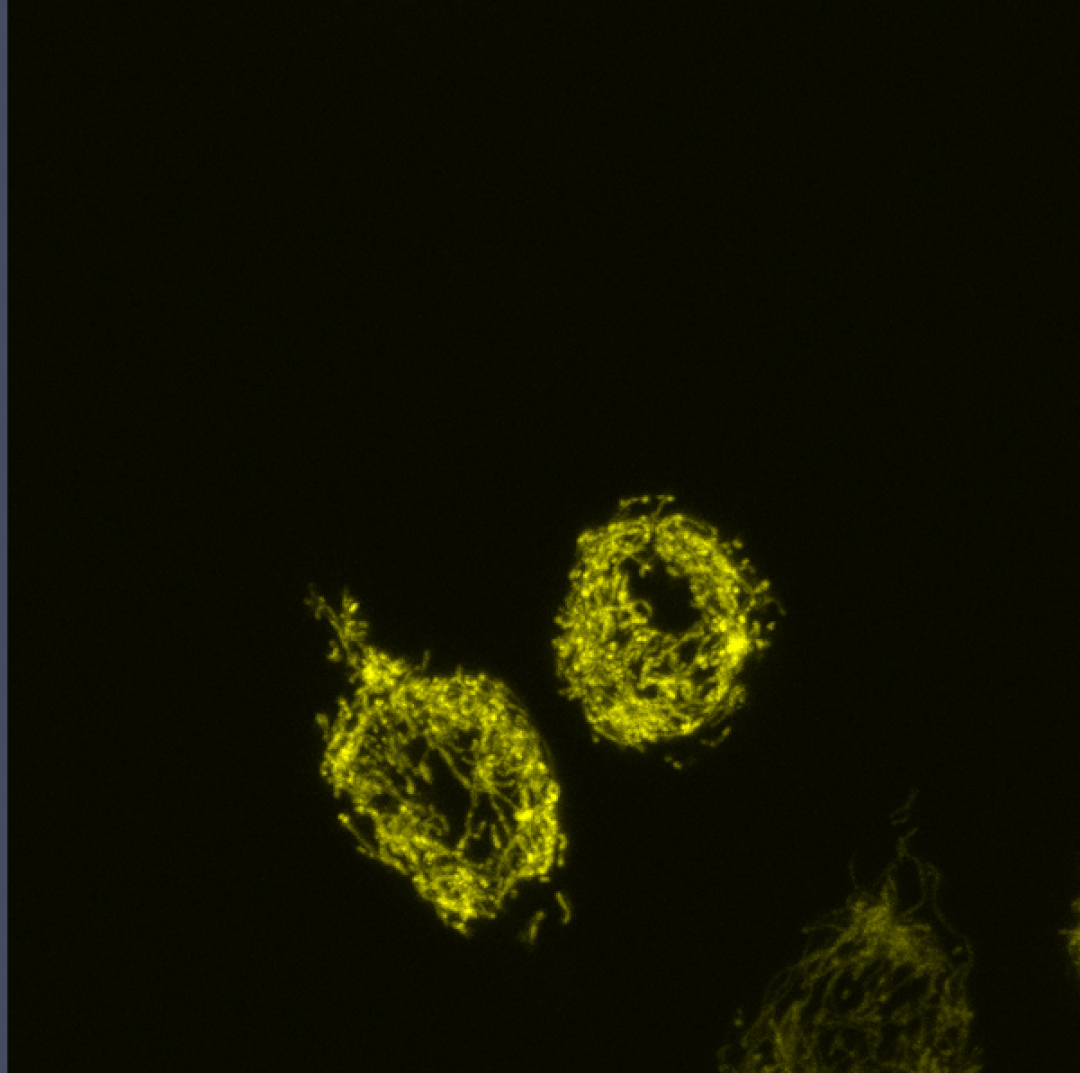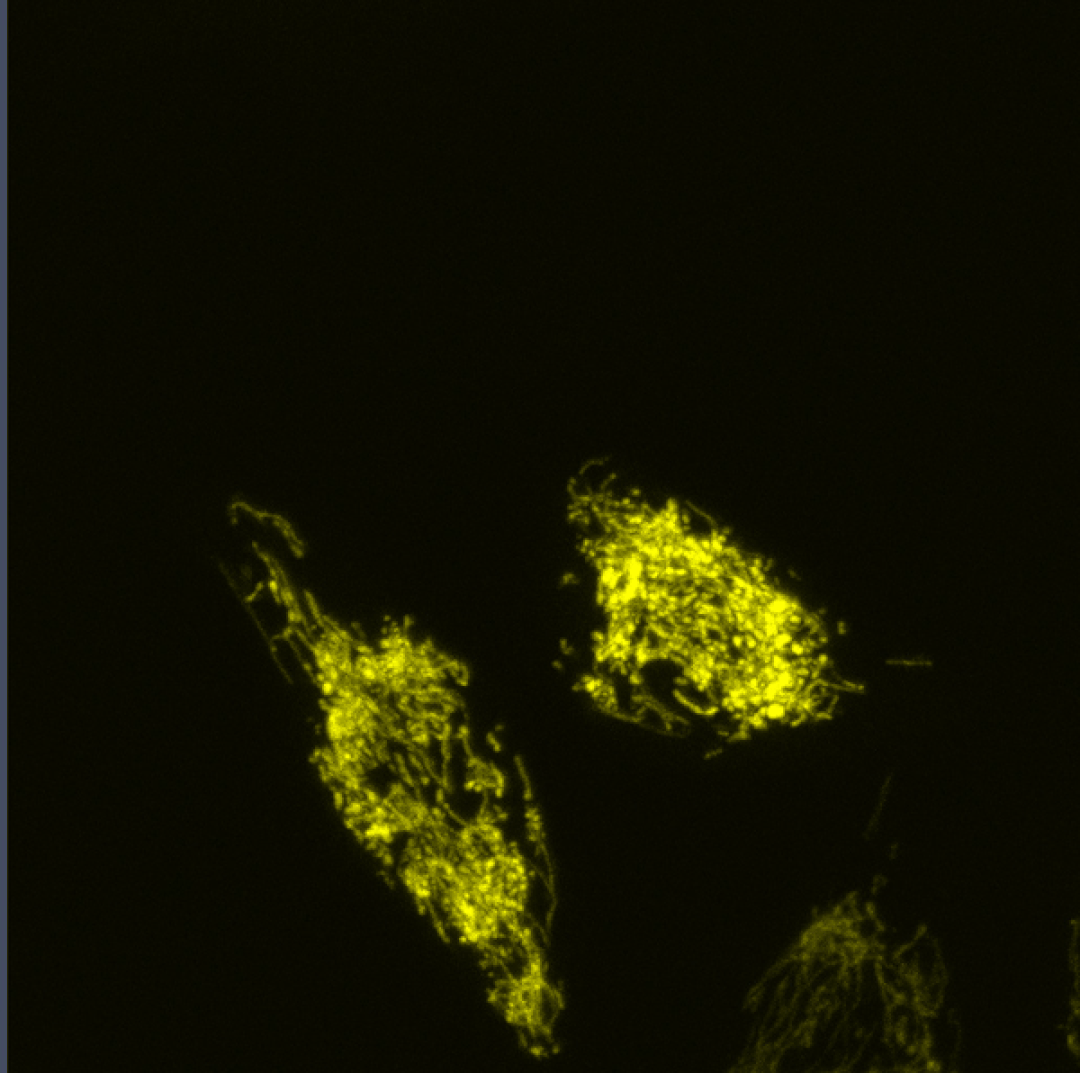
Coupling of Cell Cycle Kinetics and Mitochondrial Energetics
We seek to better understand the precise requirements for mitochondrial activity in proliferating cells during development.
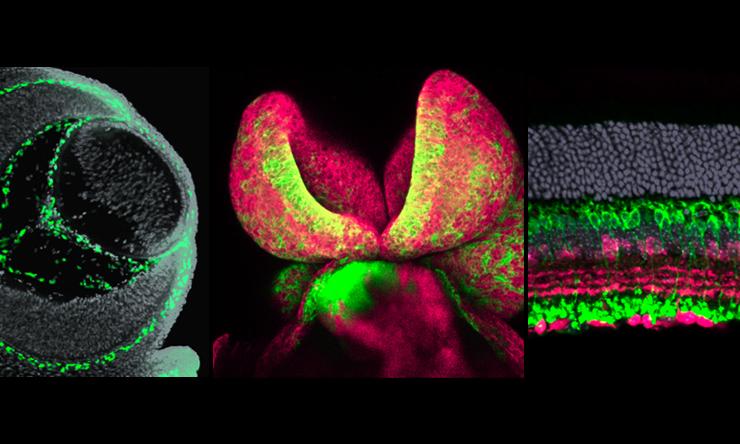
Retinal Progenitor Cell Proliferation
Our main research focus is to elucidate those transcriptional and epigenetic programs that are essential for balancing retinal progenitor cell proliferation and differentiation.
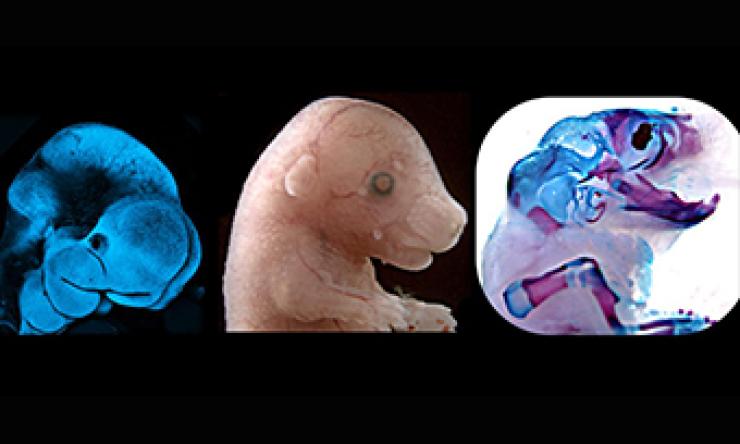
Novel Mouse Models
We have developed novel mouse models of newly discovered human syndromes impacting craniofacial and CNS development.
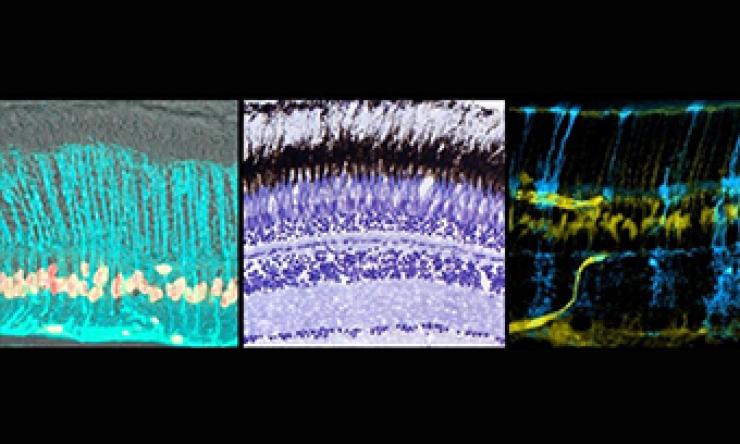
Mechanisms of Retinal Regeneration
We are currently investigating the cellular and molecular pathways negatively regulating mouse Müller glia-mediated retina regeneration.
Publications
Dr. Ross Poché and his lab members contribute to a number of publications in notable science journals.
Support our Research
Donations are used to support our research efforts to better understand and treat retinal developmental and degenerative diseases.
