HIV-1 accessory protein helps induce metabolic defects
The wily human immunodeficiency virus (HIV-1) has another trick up its sleeve, one that can disrupt cellular metabolism and raise the risk of heart disease, diabetes and osteoporosis in patients already receiving treatment to fight HIV, said researchers led by those at Baylor College of Medicine in a report that appears online in the journal Science Translational Medicine.
The culprit is an HIV-1 accessory protein known as viral protein R (Vpr). The findings reported by the researchers answer some of the questions as to why patients receiving successful treatment for HIV end up with abnormal fat loss and deposits – a problem call lipodystrophy.
“With antiretroviral treatment, HIV has become a chronic illness, and lipodystrophy and associated metabolic defects have become, long-term, serious complications,” said Dr. Ashok Balasubramanyam, professor of medicine – endocrinology, diabetes and metabolism at BCM. “It appears that latent or hidden HIV is dangerous not only because it can flare up if treatment is interrupted, but also because it keeps producing toxic signals that make HIV disease (despite the best antiviral treatment) a chronic, metabolically debilitating illness with high risk of diabetes, cardiovascular disease and obesity. This adds to the urgent need for a real cure for HIV, i.e., ways of wiping out all traces of HIV even in reservoirs where it currently hides out.”
Because the HIV lipodystrophy syndrome was first noticed around the time that the successes of anti-retroviral drugs became apparent, most clinicians assumed that the drugs were the cause. However, research shows that while the drugs may play a role, some of the problems with lipids in the blood and glucose levels and diseases associated with those problems continue, even if the drugs are changed or newer drugs are used. Fat metabolic defects also exist in patients who are untreated or have a genetic quirk that prevents the virus from destroying their immune systems after infection.
Focus on HIV01 virus protein R
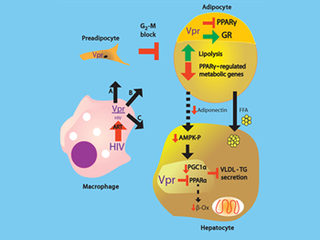
In this case, Balasubramanyam and his colleagues focused on HIV-1 virus protein R (Vpr), which can co-activate the glucocorticoid receptor and co-repress proliferator-activated receptor gamma (PPARgamma), both factors that play key roles in cellular metabolism.
“We found that Vpr circulates in the blood of patients with no detectable viral load after treatment with the best antiviral drugs,” said Balasubramanyam. “This implies that ‘latent’ or ‘persistent’ HIV that is hiding out in reservoirs is constantly turning out Vpr, which can cause the metabolic defects.”
“It appears to work as a toxic hormone that is sent out from infected cells to attack others than cannot be infected directly by HIV-1,” he said. This protein can easily infiltrate fat and liver cells, where it disrupts some of the genetic machinery and regulating proteins crucial to metabolic function.
“These metabolic pathways get disrupted, resulting in excessive breakdown of fat stores, increased release of fats into the bloodstream, and insulin resistance,” he said.
In studies in mice, he and his colleagues, including first authors Dr. Neeti Agarwal, a post-doctoral associate, and Dr. Dinakar Iyer, an instructor in the Department of Medicine – Endocrinology, Diabetes and Metabolism, both at BCM, found that mice that produce Vpr in the liver and fat cells suffer disruption of metabolic pathways. In another experiment, they pumped Vpr in mice as though it were a drug to recreate what happens in patients. Again, the metabolic pathways were disturbed.
“One big surprise was how closely the specific defects in each model resembled the defects in the other – proof that not only is Vpr capable of disrupting these metabolic pathways, but can do so when introduced into the bloodstream of the mice. The second was the finding of fatty liver in these mice. Fatty liver is very prevalent in HIV patients, but we weren’t expecting it in these mice,” said Balasubramanyam. They found that Vpr interferes with pathways involving proper handling of fat by the liver.
Possible uses
Addressing the mechanisms mediated by Vpr might help HIV patients with the complications of insulin resistance, fatty liver, diabetes and heart disease, he said. One possibility might be using a drug or neutralizing antibodies to block the overall effect of Vpr. It might also be possible to use a drug such as mifepristone to block the glucocorticoid receptor, which Vpr activates.
“These findings add to the urgent need for a real cure for HIV, wiping out all traces of the virus, even in reservoirs where it hides,” he said.
It also suggests areas for further research with viruses, particularly those that are chronic or hide in the body for long periods, such as herpes, adenovirus or cytomegalovirus (CMV).
Revise thinking
“It would be a good idea to focus research efforts on uncovering more connections between chronic infections and metabolic diseases. Hitherto infectious diseases and metabolic diseases have been considered to be separate and unconnected – I think it’s time to revise our thinking about that,” he said.
Others who took part in this research include: Rajagopal V. Sekhar, Toni Oplt, Eric D. Buras, Susan L. Samson, Maria C. Rodriguez-Barradas and Farook Jahoor, all of BCM; Terry M. Phillips and Tomoshige Kino, both of the National Institutes of Health; Ulrich Schubert of the University of Erlangen in Germany; Jacob Couturier and Dorothy E. Lewis, both of The University of Texas Health Science Center at Houston; Jeffrey B. Kopp of the National Institute of Diabetes and Digestive and Kidney Diseases; and Sanjeet G. Patel of the University of California Los Angeles Medical Center in California. Balasubramanyam, Sekhar and Samson are also with Harris Health System’s Ben Taub Hospital. Rodriguez-Barradas is also with the Michael E. DeBakey Veterans Affairs Medical Center. Buras is now with the University of Michigan in Ann Arbor.
This work was supported by the National Institute of Diabetes and Digestive and Kidney Diseases (Grant DK081553) and a Developmental Grant from the Baylor Center for AIDS Research (National Institute of Allergy and Infectious Diseases [Grant P30AI36211]), a Seed award (NIDDK Grant P30DK079638 [Diabetes Research Center at Baylor]), German Research Council (Grants SFB 796 and 643), the NIDDK Intramural Research Program, and the NIH Office of the Director.
Read more about research at Baylor College of Medicine in our monthly newsletter From the Labs.


