Recent work has shown us that G3BP nucleation of stress granules in the absence of virus infection activates PKR, resulting in phosphorylation of eIF2alpha and activation of NF-kB and JNK signal pathways. We have also shown that G3BP-induced stress granules in the uninfected cell system can nucleate several downstream interferon activated signaling and effector molecules such as RNaseL, and OAS2. We are testing the hypothesis that stress granules are staging platforms that activate or amplify downstream innate immune signaling. This could be a major function of stress granules that all viruses must counteract to effectively replicate.
We have also shown that expression of the P-body component Dcp1a results in activation of PKR that leads to translation restriction. This indicates that a new interface exists between RNA decay proteins (Dcp1a) that exist in P-bodies as well. Together, these results suggest that stress mobilization of stress granules and increased P-bodies signals with innate immune pathways to form an integrated stress response that senses virus infection in new ways.
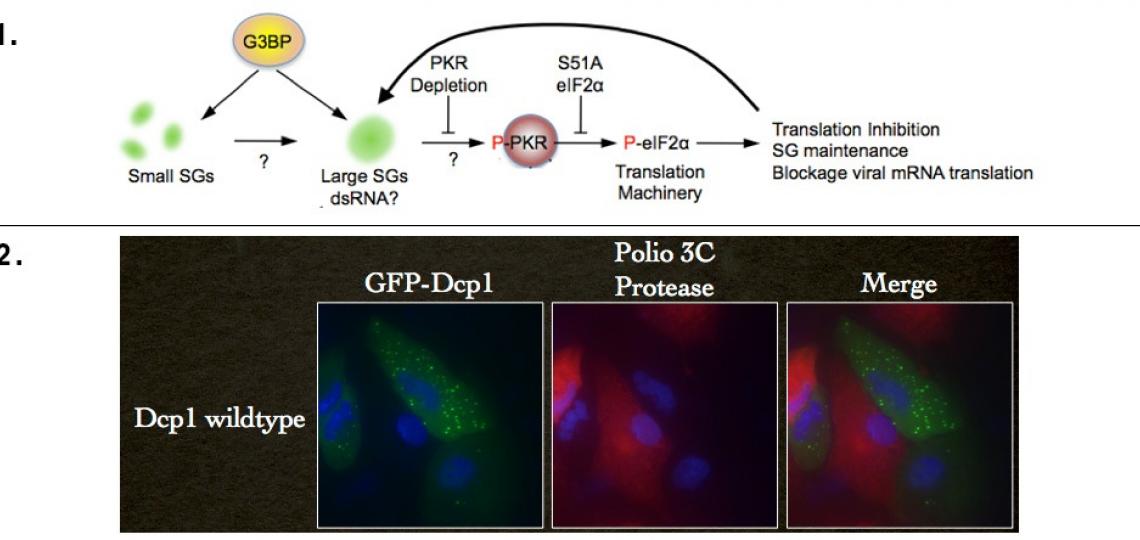
Top (1): Scheme illustrating assembly of large stress granules activating PKR.
Bottom (2): Expression of Dcp1a induces P-body formation (green foci) but also PKR activation, resulting in blockage of poliovirus replication (absence of 3C protease stain in cells expressing Dcp1a).







